
Germanium oxide (GeO 2) is basically used in camera lenses, fibre optics, microscopes as well as infrared optics. Let me explain this with a simple example by using the concept of conduction band and valence band. Hence, Germanium is called semiconductor. It has the conductivity which is higher than nonmetals, but lower than metals. Germanium is called semiconductor because it is not a good conductor like metals and also not a bad conductor like nonmetals. How many Metalloids are on periodic table? Why Germanium is called Semiconductor? 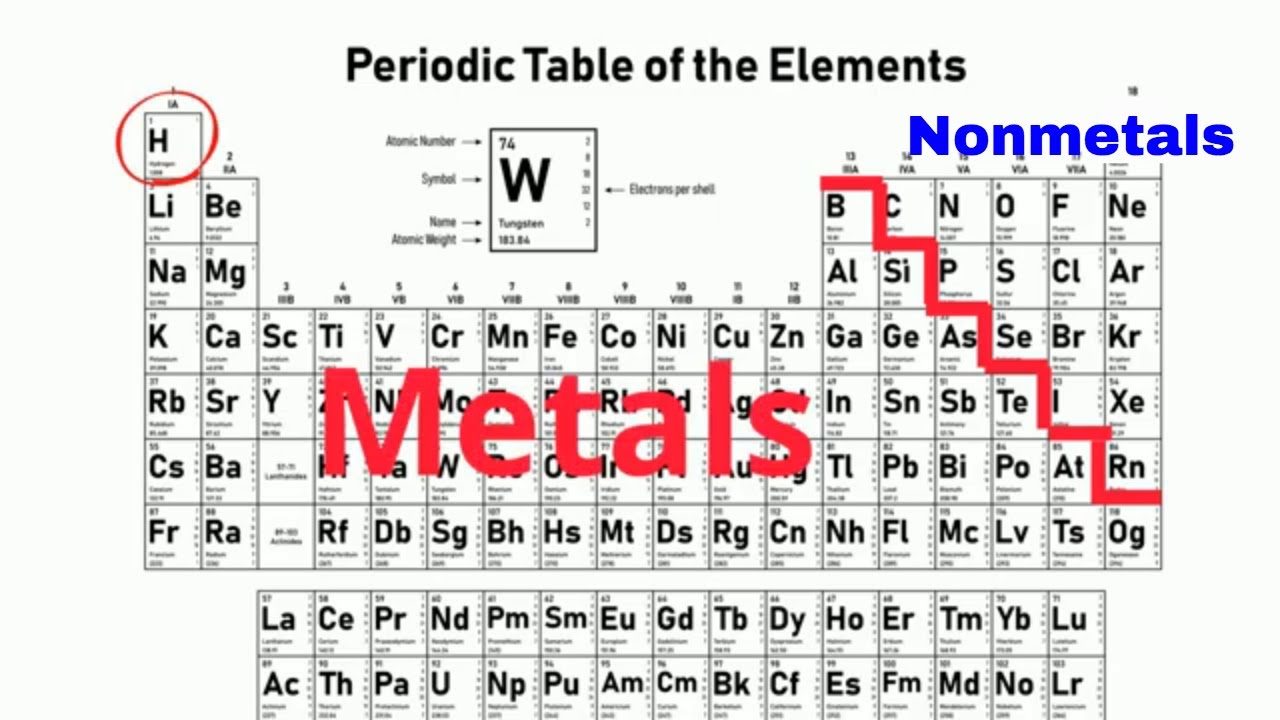
Metals, Nonmetals and Metalloids on periodic table (Image)Ģ). Hence, as germanium shows properties of both metals as well as non metals, it is classified as metalloid.ġ). It is neither hard like metals nor soft like nonmetals, but it is brittle in nature. It is neither a good conductor of electricity like metals nor a bad conductor of electricity like nonmetals. Germanium element has a metallic luster but it is not a metal. In other words, Germanium shows properties that are a mixture of metallic properties as well as nonmetallic properties. It shows few traits of metals as well as few traits of nonmetals. Germanium is a Metalloid (that means it shows properties of both metals as well as nonmetals.) Protons 32 Neutrons 41 Electrons 32 Symbol Ge Atomic massĢ, 8, 18, 4 Electronic configuration 3d 10 4s 2 4p 2 Atomic radiusĢ11 picometers (van der Waals radius) Valence electronsĤ 1st Ionization energy 7.9 eV ElectronegativityĭC (Diamond cubic) Melting point 1211.40 K or 938.2 ☌ or 1720.8 ☏ Boiling point 3106 K or 2833 ☌ or 5131 ☏ Density 5.323 g/cm 3 Main isotope 74Ge Who discovered Germanium and when? Grayish white State (at STP) Solid Position in Periodic table Germanium Element (Ge) Information Appearance So if you want to know anything about Germanium element, then this guide is for you. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Germanium element in Periodic table.) This is a SUPER easy guide on Germanium element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
